

Course Work BY R. A. Chaproniere
Navigation
HOMEPAGE
COURSES
![]() Exploring the Universe
Exploring the Universe
![]() Thu' a Small Telescope
Thu' a Small Telescope
![]() Thu' a Large Telescope
Thu' a Large Telescope
![]() Galaxies
Galaxies
![]() Planetary Atmospheres
Planetary Atmospheres
![]() Planetary Geology
Planetary Geology
![]() Modern Cosmology
Modern Cosmology
![]() Stars
Stars
![]() Science Fiction???
Science Fiction???
PROJECTS
![]() Essays
Essays
![]() Observational Work
Observational Work
ABOUT ME
CONTACT ME ![]()
Data Source :
Star data from the Stars web-site Graphic from the Astralia telescope web-site
Advertisement
 Hubble Reborn takes the reader on a journey through the Universe with spectacular full-colour pictures of galaxies, nebulae, planets and stars as seen through Hubble's eyes, along the way telling the dramatic story of the space telescope, including interviews with key scientists and astronauts.
Hubble Reborn takes the reader on a journey through the Universe with spectacular full-colour pictures of galaxies, nebulae, planets and stars as seen through Hubble's eyes, along the way telling the dramatic story of the space telescope, including interviews with key scientists and astronauts.
Astonomy Now
![]()
Advertisement
John Moores University offers exciting distance learning courses in Astronomy ranging from an Introduction to Astronomy to Planetary Geology . You are sure to find something that interests you and want to know more about. If, like me, you've a lifelong interest in the subject or have been overwhelmed by the sheer beauty of the heavens then this is the place to start.
John Moores University
![]()
Assignment 4 (Energy Flow from the Sun)
![]()
To See Other Assignments in the Exploring the Universe Series: | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
Energy Flow from the Sun - Continued
Around the year 1857 Robert Bunsen inventor of the gas burner named after him discovered together with his colleague and fellow scientist Gustav Kirchhoff that when light from a flame contaminated with a particular element passed through a prism it produced coloured bands of light ona dark background. These bands of light were peculiar to that element and that element alone. Kirchhoff went on in the early 1860's to formulate three laws that are named in his honour that describe the properties of both Absorption and Emission spectra.
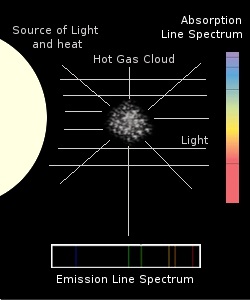 What determines the difference between an absorption and an emission spectrum has to do with the arrangement of the various features producing it. After the first law which describes how a perfect spectrum with no lines is produced his second law goes on to explain that where the heating and illuminating source is not behind the gas the elemental atoms radiate in their particular wavelength producing the bright spectral lines of emission. The third law states that where the heat source is optically in line with and behind the gas in question then the elemental atoms absorb their particular wave lengths producing an absorption spectrum.
What determines the difference between an absorption and an emission spectrum has to do with the arrangement of the various features producing it. After the first law which describes how a perfect spectrum with no lines is produced his second law goes on to explain that where the heating and illuminating source is not behind the gas the elemental atoms radiate in their particular wavelength producing the bright spectral lines of emission. The third law states that where the heat source is optically in line with and behind the gas in question then the elemental atoms absorb their particular wave lengths producing an absorption spectrum.
An atom absorbing light at specific wavelengths and then re-radiating them does so in any direction. This means that, with no back lighting our point of observation must encounter some of that radiation. Set against a dark background this emission must appear bright.
 Moving our point of observation so that the heat source is behind the atom means that although it still absorbs and re-radiates at specific wavelengths it still does so in any direction. With some wavelengths streaming towards us uninhibited and others being absorbed and re-radiated, some back in the direction of the light source those spectral lines will appear dark against a bright background. A rough analogy of this are Sun Spots. Although they appear dark on the surface of the Sun they would appear very bright if the were on their own.
Moving our point of observation so that the heat source is behind the atom means that although it still absorbs and re-radiates at specific wavelengths it still does so in any direction. With some wavelengths streaming towards us uninhibited and others being absorbed and re-radiated, some back in the direction of the light source those spectral lines will appear dark against a bright background. A rough analogy of this are Sun Spots. Although they appear dark on the surface of the Sun they would appear very bright if the were on their own.
Pages
<< | Previous | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | >>